Flubendazole
In today's article, we will explore Flubendazole and its impact on today's world in detail. From its origin to its evolution today, Flubendazole has played a crucial role in different aspects of daily life. Over the years, Flubendazole has sparked great interest and debate, generating multiple perspectives and opinions that have shaped its meaning and relevance in society. From his impact on popular culture to his influence on technology and science, Flubendazole has left an indelible mark on the contemporary landscape. In this article, we will explore how Flubendazole has shaped the world we live in and what its implications are for the future.
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| License data |
|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.046.007 |
| Chemical and physical data | |
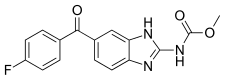
| Formula | C16H12FN3O3 |
| Molar mass | 313.288 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 243 °C (469 °F) |
| Solubility in water | 10.0 ± 0.4 × 10−6 μg/mL (pH 1.6); 0.29 ± 0.06 × 10−6 μg/mL (pH 6.5) mg/mL (20 °C) |
| |
| |
| | |
Flubendazole is an anthelmintic, used both in humans and for veterinarian purposes. It is very close chemically to mebendazole, the only difference being an added fluorine group.
Human use
It is available for human use to treat worm infections. In certain countries such as France, it is inexpensive and available OTC (without prescription) under the brand name Fluvermal as an alternative to mebendazole which is not currently sold there.
Veterinarian use
Under veterinary use, its brand name is Flutelmium which is a paste manufactured by Janssen Pharmaceutica N.V. used by veterinarians for protection against internal parasites and worms in dogs and cats. Other brand names are Flubenol, Biovermin, and Flumoxal.
Since 2000, Flubendazole-treated grit has increasingly been laid out on a landscape-scale across many UK grouse-shooting moors by gamekeepers in an attempt to reduce the impact on bird numbers from strongyle worm. Evidence of high worm burden is required before a veterinarian can dispense and sell the product, known as 'medicated grit'. However, there has been increasing concern about contaminants entering the ground waters running off from moorlands, as well as from its use in farming environments and its presence in manure. Researchers are starting to gather research evidence in order to inform policy development on the presence of this and other veterinary medicines in the wider environment.
References
- ^ a b Vasilev NA, Voronin AP, Surov AO, Perlovich GL (March 2023). "Influence of Co-amorphization on the Physical Stability and Dissolution Performance of an Anthelmintic Drug Flubendazole". Molecular Pharmaceutics. 20 (3): 1657–1669. doi:10.1021/acs.molpharmaceut.2c00873. PMID 36732935. S2CID 256546280.
- ^ Heyer F, Tourte-Schaeffer C, Ancelle T, Faurant C, Lapierre J (1 February 1982). "Le flubendazole : un progrès dans le traitement des helminthiases intestinales. A propos de 471 observations". Médecine et Maladies Infectieuses (in French). 12 (2): 57–61. doi:10.1016/S0399-077X(82)80047-4. ISSN 0399-077X.
- ^ "FLUVERMAL". VIDAL (in French). Retrieved 31 January 2023.
- ^ "VERMOX". VIDAL (in French).
- ^ US 5824336, Jozef EM, Gilis JP, "Chewable flubendazole tablets for companion animals", issued 20 October 1998, assigned to Jannsen Pharmaceuticals
- ^ "Best practice use of medicated grit". Game and Wildlife Conservation Trust. Retrieved 27 September 2016.
- ^ Kay P. "Analysis, occurrence and effects of flubendazole in moorland river catchments". www.nercdtp.leeds.ac.uk. Leeds York NERC Doctoral Training Partnership (DTP). Retrieved 11 January 2017.
- ^ Kreuzig R, Blümlein K, Höltge S (November 2007). "Fate of the Benzimidazole Antiparasitics Flubendazole and Fenbendazole in Manure and Manured Soils". CLEAN – Soil, Air, Water. 35 (5): 488–494. Bibcode:2007CSAW...35..488K. doi:10.1002/clen.200720023. Retrieved 11 January 2017.