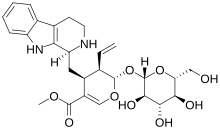
Strictosidine
In today's world, Strictosidine has become a topic of interest to a wide variety of people. With the passage of time, Strictosidine has gained relevance in different areas, whether in the personal, academic, professional, political or cultural sphere. The importance of Strictosidine is not limited to a single area, but its impact extends to multiple aspects of daily life. In this article, we will explore the different facets of Strictosidine and its influence on today's society, analyzing both its positive aspects and its challenges. Through a multidisciplinary approach, we seek to deepen the understanding of Strictosidine and its meaning in the contemporary world.
| |
| Names | |
|---|---|
| IUPAC name
Methyl (19S,20R)-19-(β-D-glucopyranosyloxy)-16,17,21,21a-tetradehydro-18-oxa-21a-homo-20,21-secoyohimban-16-carboxylate
| |
| Systematic IUPAC name
Methyl (4S,5R,6S)-5-ethenyl-4-{indol-1-yl]methyl}-6-{oxy}-5,6-dihydro-4H-pyran-3-carboxylate | |
| Other names
Isovincoside
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C27H34N2O9 | |
| Molar mass | 530.574 g·mol−1 |
| Melting point | 193-197 °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Strictosidine is a natural chemical compound and is classified as a glucoalkaloid and a vinca alkaloid. It is formed by the Pictet–Spengler condensation reaction of tryptamine with secologanin, catalyzed by the enzyme strictosidine synthase. Thousands of strictosidine derivatives are sometimes referred to by the broad phrase of monoterpene indole alkaloids. Strictosidine is an intermediate in the biosynthesis of numerous pharmaceutically valuable metabolites including quinine, camptothecin, ajmalicine, serpentine, vinblastine, vincristine and mitragynine.
Biosynthetic pathways help to define the subgroups of strictosidine derivatives.
Distribution
Strictosidine is found in the following plant families:
Here especially in Rhazya stricta and Catharanthus roseus.
Recent efforts in metabolic engineering have permitted the synthesis of strictosidine by yeast (Saccharomyces cerevisiae). This was accomplished by adding 21 genes and 3 gene deletions.
Research
The involvement of the glucoalkaloid strictosidine in the antimicrobial and antifeedant activity of Catharanthus roseus leaves was studied. Strictosidine and its deglucosylation product, specifically formed by the enzyme strictosidine glucosidase, were found to be active against several microorganisms.
References
- ^ Mizukami, H; Nordlöv, H; Lee, S. L.; Scott, A. I. (1979). "Purification and properties of strictosidine synthetase (an enzyme condensing tryptamine and secologanin) from Catharanthus roseus cultured cells". Biochemistry. 18 (17): 3760–3. doi:10.1021/bi00584a018. PMID 476085.
- ^ Treimer, J. F.; Zenk, M. H. (1979). "Purification and properties of strictosidine synthase, the key enzyme in indole alkaloid formation". European Journal of Biochemistry. 101 (1): 225–33. doi:10.1111/j.1432-1033.1979.tb04235.x. PMID 510306.
- ^ David S Seigler (1998). Plant Secondary Metabolism. Springer.
- ^ Michael Wink (2010). Biochemistry of Plant Secondary Metabolism. Blackwell.
- ^ Brown, S; Clastre, M; Courdavault, V; O'Connor, S. E. (2015). "De novo production of the plant-derived alkaloid strictosidine in yeast". Proceedings of the National Academy of Sciences. 112 (11): 3205–3210. Bibcode:2015PNAS..112.3205B. doi:10.1073/pnas.1423555112. PMC 4371906. PMID 25675512.
- ^ Luijendijk, T. J. C., van der Meijden, E. & Verpoorte, R. (1996). Involvement of strictosidine as a defensive chemical inCatharanthus roseus. Journal of Chemical Ecology, 22(8), 1355–1366. https://doi.org/10.1007/bf02027718