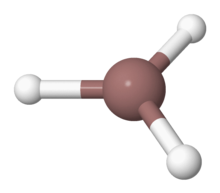
Indium trihydride
In today's world, _Indium trihydride__ has become a topic of great relevance and interest to a wide spectrum of people. Whether _Indium trihydride__ is an iconic figure, a fundamental concept or a significant date, its importance transcends borders and cultures. In this article, we will explore the different aspects related to _Indium trihydride__, from its impact on society to its influence on history. Through detailed analysis, we will seek to understand how _Indium trihydride__ has shaped our world and remains relevant today. This article aims to provide a comprehensive and enriching vision of _Indium trihydride__, inviting the reader to reflect and delve into its meaning and relevance in everyday life.
| |
| Names | |
|---|---|
| Systematic IUPAC name | |
| Other names
Indium(III) hydride
Indium trihydride | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| 163932 | |
PubChem CID
|
|
| |
| Properties | |
| InH3 | |
| Molar mass | 117.842 g/mol |
| Melting point | −90 °C (−130 °F; 183 K) (decomposes) |
| Structure | |
| Trigonal planar | |
| Dihedral | |
| Related compounds | |
Related metallanes
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Indium trihydride is an inorganic compound with the chemical formula (InH3). It has been observed in matrix isolation and laser ablation experiments. Gas phase stability has been predicted. The infrared spectrum was obtained in the gas phase by laser ablation of indium in presence of hydrogen gas InH3 is of no practical importance.
Chemical properties
Solid InH3 is a three-dimensional network polymeric structure, where In atoms are connected by In-H-In bridging bonds, is suggested to account for the growth of broad infrared bands when samples of InH3 and InD3 produced on a solid hydrogen matrix are warmed. Such a structure is known for solid AlH3. When heated above −90 °C, indium trihydride decomposes to produce indium–hydrogen alloy and elemental hydrogen. As of 2013, the only known method of synthesising indium trihydride is the autopolymerisation of indane below −90 °C.
Other indium hydrides
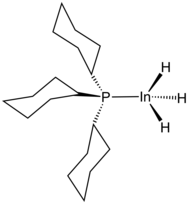
Several compounds with In-H bonds have been reported. Examples of complexes with two hydride ligands replaced by other ligands are (K+)3[K((CH3)2SiO)+7][InH(CH2C(CH3)3)−3]4 and HIn(−C6H4−ortho-CH2N(CH3)2)2.
Although InH3 is labile, adducts are known with the stoichiometry InH3Ln (n = 1 or 2). 1:1 amine adducts are made by the reaction of Li+[InH4]− (lithium tetrahydridoindate(III)) with a trialkylammonium salt. The trimethylamine complex is only stable below −30 °C or in dilute solution. The 1:1 and 1:2 complexes with tricyclohexylphosphine (PCy3) have been characterised crystallographically. The average In-H bond length is 168 pm. Indium hydride is also known to form adducts with NHCs.
References
- ^ a b "Indigane (CHEBI:30429)". Chemical Entities of Biological Interest (ChEBI). UK: European Bioinformatics Institute.
- ^ Pullumbi, P.; Bouteiller, Y.; Manceron, L.; Mijoule, C. (July 1994). "Aluminium, gallium and indium trihydrides. An IR matrix isolation and ab initio study". Chemical Physics. 185 (1): 25–37. Bibcode:1994CP....185...25P. doi:10.1016/0301-0104(94)00111-1.
- ^ Aldridge, S.; Downs, A. J. (2001). "Hydrides of the Main-Group Metals: New Variations on an Old Theme". Chemical Reviews. 101 (11): 3305–65. doi:10.1021/cr960151d. PMID 11840988.
- ^ Hunt, P.; Schwerdtfeger, P. (1996). "Are the Compounds InH3 and TlH3 Stable Gas Phase or Solid State Species?". Inorganic Chemistry. 35 (7): 2085–2088. doi:10.1021/ic950411u.
- ^ a b Andrews, L.; Wang, X. (2004). "Infrared Spectra of Indium Hydrides in Solid Hydrogen and of Solid Indane". Angewandte Chemie International Edition. 43 (13): 1706–1709. doi:10.1002/anie.200353216. PMID 15038043.
- ^ Turley, J. W.; Rinn, H. W. (1969). "The Crystal Structure of Aluminum Hydride". Inorganic Chemistry. 8 (1): 18–22. doi:10.1021/ic50071a005.
- ^ a b c Jones, C. (2001). "The stabilisation and reactivity of indium trihydride complexes". Chemical Communications (22): 2293–2298. doi:10.1039/b107285b. ISSN 1359-7345. PMID 12240044.
- ^ Rowen Churchill, M.; Lake, C. H.; Chao, S.-H. L.; Beachley, O. T. (1993). "Silicone grease as a precursor to a pseudo crown ether ligand: crystal structure of 34". Journal of the Chemical Society, Chemical Communications. 1993 (20): 1577–1578. doi:10.1039/C39930001577.
- ^ Wang, X.; Andrews, L. (20 May 2004). "Infrared Spectra of Indium Hydrides in Solid Hydrogen and Neon". The Journal of Physical Chemistry A. 108 (20): 4440–4448. Bibcode:2004JPCA..108.4440W. doi:10.1021/jp037942l.
- ^ Abernethy, C. D.; Cole, M. L.; Jones, C. (2000). "Preparation, Characterization, and Reactivity of the Stable Indium Trihydride Complex ". Organometallics. 19 (23): 4852–4857. doi:10.1021/om0004951.