Mipafox
In this article we will explore Mipafox in depth, a topic that has sparked great interest and debate in recent years. From its origins to its relevance today, Mipafox has captured the attention of various spheres of society. Through an exhaustive analysis, we will examine the different edges that make up Mipafox, as well as their implications in different contexts. In addition, we will emphasize the evolution of Mipafox over time, highlighting its impact in different areas and its influence on decision making. By weighing the various points of view on Mipafox, we seek to provide a comprehensive perspective that enriches the understanding of this topic that is so relevant today.
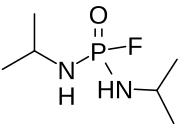
| |
| Names | |
|---|---|
| Preferred IUPAC name
N,N′-Di(propan-2-yl)phosphorodiamidic fluoride | |
| Other names
Bis(isopropylamino)fluorophosphine oxide; Isopestox
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.006.130 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H16FN2OP | |
| Molar mass | 182.179 g·mol−1 |
| Density | 1.2 |
| Melting point | 65 °C (149 °F; 338 K) |
| Boiling point | 125 °C (257 °F; 398 K) |
| 80 g/L | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Highly toxic |
| GHS labelling: | |
 
| |
| Danger | |
| H301, H370 | |
| P260, P264, P270, P301+P310, P307+P311, P321, P330, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Mipafox is a highly toxic organophosphate insecticide that is an irreversible acetylcholinesterase inhibitor and is resistant to cholinesterase reactivators. It was developed in the 1950s and is now believed to be no longer in use.
Toxicity
There are case reports of delayed neurotoxicity and paralysis due to acute exposure to mipafox.
Synthesis
Phosphoryl chloride is first reacted with isopropylamine. The resulting product is then reacted with potassium fluoride or ammonium fluoride to produce mipafox.
See also
References
- ^ Mangas, I; Taylor, P; Vilanova, E; Estévez, J; França, TC; Komives, E; Radić, Z (March 2016). "Resolving pathways of interaction of mipafox and a sarin analog with human acetylcholinesterase by kinetics, mass spectrometry and molecular modeling approaches". Archives of Toxicology. 90 (3): 603–16. doi:10.1007/s00204-015-1481-1. PMC 4833118. PMID 25743373.
- ^ "The WHO Recommended Classification of Pesticides by Hazard and Guidelines to Classification 2019" (PDF). World Health Organization.
- ^ BIDSTRUP, PL; BONNELL, JA; BECKETT, AG (16 May 1953). "Paralysis following poisoning by a new organic phosphorus insecticide (mipafox); report on two cases". British Medical Journal. 1 (4819): 1068–72. doi:10.1136/bmj.1.4819.1068. PMC 2016450. PMID 13042137.
- ^ "Process for the preparation of bisisopropyl-amino-fluoro-phosphine oxide".