A-262
In today's world, A-262 has become a topic of great relevance and interest. Whether due to its impact on society, its importance in history, its influence on popular culture or any other aspect that makes it stand out, A-262 is a topic that deserves to be explored in depth. In this article, we will dive into the fascinating world of A-262, analyzing its various aspects and discovering its meaning in the current context. From its origin to its relevance today, A-262 is a topic that never ceases to intrigue experts and fans alike. Join us on this journey of discovery and learning about A-262.
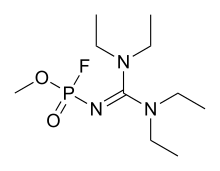
| |
| Names | |
|---|---|
| IUPAC name
1,1,3,3-tetraethyl-2-guanidine
| |
| Identifiers | |
3D model (JSmol)
|
|
PubChem CID
|
|
| |
| |
| Properties | |
| C10H23FN3O2P | |
| Molar mass | 267.285 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
A-262 is an organophosphate nerve agent. It was developed in the Soviet Union under the FOLIANT program and is one of the group of compounds referred to as Novichok agents that were revealed by Vil Mirzayanov. Mirzayanov gives little specific information about A-262, stating that it is highly toxic, but no figures are given to compare it to other related agents. It is reportedly a solid rather than a volatile liquid as with most nerve agents, and in order to weaponise it successfully, it had to be milled into a fine powder form that could be dispersed as a dust.
Legal status
A-262 is not specifically listed in the Annex on Chemicals of the Chemical Weapons Convention, and falls outside the general definitions covering other Novichok agents, even though it has a chemical structure similar to that of both A-232 (the example compound for schedule 1.A.14) and A-242 (the example compound for schedule 1.A.15). It would however be considered a "Toxic Chemical" under the catch-all provisions due to its potent acetylcholinesterase inhibitor activity and potential military applications, but the level of control it is subject to is lower than that for compounds specifically listed under Schedule 1.
See also
References
- ^ Mirzayanov VS (2008). State Secrets: An Insider's Chronicle of the Russian Chemical Weapons Program. Outskirts Press. ISBN 978-1-4327-2566-2.
- ^ "Guidelines for Schedules of Chemicals". Organisation for the Prohibition of Chemical Weapons (OPCW). Retrieved 2020-10-07.