Balovaptan
Nowadays, Balovaptan is a topic on everyone's lips. With the advancement of technology and globalization, Balovaptan has become a constant concern for modern society. Whether in the political, economic, social or environmental sphere, Balovaptan has generated debate and sparked the interest of experts and citizens alike. In this article, we will explore the different facets of Balovaptan and analyze its impact on our lives. From its origin to its possible solutions, we will reflect on how Balovaptan influences our daily lives and what we can do about it.
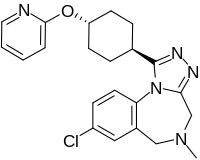 | |
| Clinical data | |
|---|---|
| Other names | RG7314 |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C22H24ClN5O |
| Molar mass | 409.92 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Balovaptan (INN; developmental code name RG7314), is a selective small molecule antagonist of the vasopressin V1A receptor which is under development by Roche for the treatment of post-traumatic stress disorder.
Clinical studies
Post-traumatic stress disorder
It was in a phase III clinical trial for adults and a phase II clinical trial for children for this indication.
Autism
In January 2018, Roche announced that the US Food and Drug Administration (FDA) had granted breakthrough therapy designation for balovaptan in people with autism spectrum disorder (ASD). The FDA granted this based on the results of the adult phase II clinical trial called VANILLA (Vasopressin ANtagonist to Improve sociaL communication in Autism) study. The phase III adult study (March 2020) is called V1aduct and the phase II child study is called Av1ation.[citation needed]
The phase III study concluded that balovaptan did not improve social communication in autistic adults.
It was also in phase II studies for the treatment of stroke. However, it has since been discontinued for both of those indications, and is only being developed for post-traumatic stress disorder, with plans to submit regulatory filings in 2025 or later.
References
- ^ "Roche - Pipeline". 2023. Retrieved 5 April 2023.
- ^ Clinical trial number NCT01793441 for "Study of RG7314 to Investigate Efficacy and Safety in Individuals With Autism Spectrum Disorders" at ClinicalTrials.gov
- ^ "FDA grants Breakthrough Therapy Designation for Roche's balovaptan in autism spectrum disorder" (Press release). 29 January 2018. Retrieved 6 February 2018.
- ^ Bolognani F, Del Valle Rubido M, Squassante L, Wandel C, Derks M, Murtagh L, et al. (May 2019). "A phase 2 clinical trial of a vasopressin V1a receptor antagonist shows improved adaptive behaviors in men with autism spectrum disorder". Science Translational Medicine. 11 (491). doi:10.1126/scitranslmed.aat7838. PMID 31043521.
- ^ Jacob S, Veenstra-VanderWeele J, Murphy D, McCracken J, Smith J, Sanders K, et al. (March 2022). "Efficacy and safety of balovaptan for socialisation and communication difficulties in autistic adults in North America and Europe: a phase 3, randomised, placebo-controlled trial". The Lancet. Psychiatry. 9 (3): 199–210. doi:10.1016/s2215-0366(21)00429-6. PMID 35151410.
- ^ "Balovaptan". Adis Insight. Springer Nature Switzerland AG. Retrieved 5 April 2023.