Safranal
In this article, we will explore the fascinating world of Safranal. From its origin to its impact on today's society, we will delve into its many facets and discover its influence in different areas. Safranal has been the subject of study and debate throughout history, awakening the interest of specialists and amateurs alike. Along these lines, we will examine its origins, evolution and its relevance in the current context, offering a complete and detailed vision of Safranal. Likewise, we will analyze its impact in different areas, highlighting its role in culture, technology, politics and much more. Get ready to embark on a fascinating journey through Safranal!
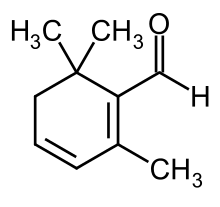
| |
| Names | |
|---|---|
| Preferred IUPAC name
2,6,6-Trimethylcyclohexa-1,3-diene-1-carbaldehyde | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.003.758 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H14O | |
| Molar mass | 150.21 g/mol |
| Density | 0.9734 g/cm3 |
| Boiling point | 70 °C (158 °F; 343 K) at 1 mmHg |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Safranal is an organic compound isolated from saffron, the spice consisting of the stigmas of crocus flowers (Crocus sativus). It is the constituent primarily responsible for the aroma of saffron.
It is believed that safranal is a degradation product of the carotenoid zeaxanthin via the intermediate picrocrocin.
Pharmacology
Safranal is an effective anticonvulsant in animal models, shown to act as an agonist at GABAA receptors. Safranal also exhibits high antioxidant and free radical scavenging activity, along with cytotoxicity towards cancer cells in vitro. One of its anticancer mechanisms of action involves disruption of the normal assembly dynamics of cellular microtubules. It has also been shown to have antidepressant properties in animals and pilot studies in humans.
Natural sources
Natural sources of safranal include:[unreliable source?]
- Microcystis (cyanobacterium)
- Aspalathus linearis (Rooibos)
- Camellia sinensis (Tea leaf)
- Crocus sativus (Saffron)
- Ficus carica (Fig leaf)
- Lycium chinense (Wolfberry)
- Cuminum cyminum (Cumin Seed)
- Centaurea sibthorpii
- Centaurea amanicola
- Centaurea consanguinea
- Erodium cicutarium (common stork's-bill or pinweed)
- Calycopteris floribunda (Ukshi)
- Sambucus nigra (elderberry)
- Citrus limon (lemon)
- Achillea distans
References
- ^ Hosseinzadeh H; Talebzadeh F (December 2005). "Anticonvulsant evaluation of safranal and crocin from Crocus sativus in mice". Fitoterapia. 76 (7–8): 722–4. doi:10.1016/j.fitote.2005.07.008. PMID 16253437.
- ^ Hosseinzadeh H; Sadeghnia HR (April 2007). "Protective effect of safranal on pentylenetetrazol-induced seizures in the rat: involvement of GABAergic and opioids systems". Phytomedicine. 14 (4): 256–62. doi:10.1016/j.phymed.2006.03.007. PMID 16707256.
- ^ Hosseinzadeh H; Sadeghnia HR (2005). "Safranal, a constituent of Crocus sativus (saffron), attenuated cerebral ischemia induced oxidative damage in rat hippocampus". Journal of Pharmacy & Pharmaceutical Sciences. 8 (3): 394–9. PMID 16401389.
- ^ Assimopoulou AN; Sinakos Z; Papageorgiou VP (November 2005). "Radical scavenging activity of Crocus sativus L. extract and its bioactive constituents". Phytotherapy Research. 19 (11): 997–1000. doi:10.1002/ptr.1749. PMID 16317646. S2CID 23907085.
- ^ Escribano J; Alonso GL; Coca-Prados M; Fernandez JA (February 1996). "Crocin, safranal and picrocrocin from saffron (Crocus sativus L.) inhibit the growth of human cancer cells in vitro". Cancer Letters. 100 (1–2): 23–30. doi:10.1016/0304-3835(95)04067-6. PMID 8620447.
- ^ Cheriyamundath S, Choudhary S, and Lopus M (2017) Safranal inhibits HeLa cell viability by perturbing the reassembly potential of microtubules. Phytother Res, 32, 170-173. doi: 10.1002/ptr.5938. PMID 29024138
- ^ Hosseinzadeh H; Karimi G; Niapoor M (2004). "Antidepressant effect of Crocus sativus L. stigma extracts and their constituents, crocin and safranal, in mice". Acta Horticulturae. 650 (650): 435–45. doi:10.17660/ActaHortic.2004.650.54.
- ^ Akhondzadeh S; Fallah-Pour H; Afkham K; Jamshidi AH; Khalighi-Cigaroudi F (September 2004). "Comparison of Crocus sativus L. and imipramine in the treatment of mild to moderate depression: A pilot double-blind randomized trial ISRCTN45683816". BMC Complementary and Alternative Medicine. 4: 12. doi:10.1186/1472-6882-4-12. PMC 517724. PMID 15341662.
- ^ "List of Chemicals". sun.ars-grin.gov. Retrieved 2008-03-02.
- ^ Yan JH; Tang KW; Zhong M; Deng NH (November 2002). "". Se Pu (in Chinese). 20 (6): 569–72. PMID 12683011.
- ^ a b c d e f g h Ramin Rezaee; Hossein Hosseinzadeh (January 2013). "[Safranal: From an Aromatic Natural Product to a Rewarding Pharmacological Agent]". Iranian Journal of Basic Medical Sciences. 16 (1): 12–26. PMC 3637901. PMID 23638289.